Majka Laboratory
Our lab studies how mesenchymal progenitor cells (MPC) regulate lung microvascular function and dysregulated angiogenesis as well as how these processes contribute to the development or severity of chronic lung diseases (including fibrosis, emphysema as well as pulmonary hypertension), vascular biology, Endothelial progenitor cells (EPC), and Alveolar–Capillary unit.
Lab Resources and Services
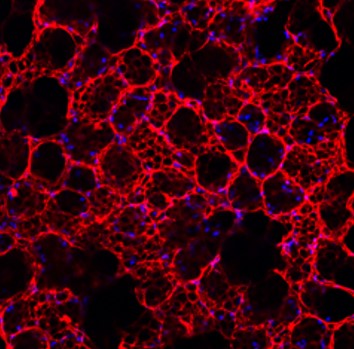 The Majka lab identified and characterized a novel population of lung-resident mesenchymal endothelial progenitor cells using the side population (SP) approach, establishing Abcg2 as a defining marker. The lab continues to use Abcg2 to isolate and study adult endogenous lung microvascular endothelial progenitor cells (mvEPCs) in both pre-clinical and human systems. These mvEPCs play critical roles in regulating angiostasis, as well as maintaining the function and structural integrity of the alveolar-capillary unit.
The Majka lab identified and characterized a novel population of lung-resident mesenchymal endothelial progenitor cells using the side population (SP) approach, establishing Abcg2 as a defining marker. The lab continues to use Abcg2 to isolate and study adult endogenous lung microvascular endothelial progenitor cells (mvEPCs) in both pre-clinical and human systems. These mvEPCs play critical roles in regulating angiostasis, as well as maintaining the function and structural integrity of the alveolar-capillary unit.
The Majka lab investigates Abcg2⁺ mvEPCs and the microvascular endothelium in the context of reparative angiogenesis and microvascular remodeling, with the goal of identifying targetable pathways that govern the transition between tissue repair and chronic lung disease. The group has developed robust techniques to isolate, differentiate, and analyze resident lung mesenchymal endothelial progenitors, examining cell phenotype, proliferation, inflammation, migration, and matrix synthesis both in vitro and in vivo using murine models and human tissue explants. Comparative studies between pre-clinical and human ABCG2⁺ lung mvEPCs have demonstrated the importance of Wnt/β-catenin and mTOR signaling in maintaining cellular function, preserving tissue structure, and promoting repair.
More recently, the Majka lab has developed advanced methods to model and quantitatively analyze the three-dimensional structure of the alveolar-capillary network using microCT and confocal imaging. Current efforts focus on understanding the heterogeneity of lung capillary endothelium and elucidating how microvascular endothelial cells and progenitors communicate with each other and with immune cells during repair processes. These innovative models position the lab to investigate how dysfunctional mvEPC activity contributes to microvascular disease mechanisms, including alveolar-capillary aging, emphysema (characterized by structural loss), fibrosis (marked by excessive proliferation), and aberrant de novo angiogenesis, all of which impact alveolar-capillary remodeling and lung function.
Current Projects

Susan M. Majka, PhD
Personnel
Principal Investigator
Susan Majka, PhD
Professor of Medicine
Division of Pulmonary, Critical Care & Sleep Medicine
Department of Biomedicl Research
National Jewish Health, Denver CO
Professor of Medicine, Division of Pulmonary & Critical Care
Gates Center for Regenerative Medicine and Stem Cell Biology
University of Colorado Health Sciences Center, Aurora CO
Opportunities
Motivated students and postdoctoral researchers are always welcome!
Publications
- Summers ME, Richmond BW, Kropski JA, Majka SA, Bastarache JA, Hatzopoulos AK, Bylund J, Ghosh M, Petrache I, Foronjy RF, Geraghty P, Majka SM. Balanced Wnt/Dickkopf-1 signaling by mesenchymal vascular progenitor cells in the microvascular niche maintains distal lung structure and function. Am J Physiol Cell Physiol. 2021 Jan 1;320(1):C119-C131. doi: 10.1152/ajpcell.00277.2020. Epub 2020 Oct 21. PubMed PMID: 33085496; PubMed Central PMCID: PMC7846975.
- Railwah C, Lora A, Zahid K, Goldenberg H, Campos M, Wyman A, Jundi B, Ploszaj M, Rivas M, Dabo A, Majka SM, Foronjy R, El Gazzar M, Geraghty P. Cigarette smoke induction of S100A9 contributes to chronic obstructive pulmonary disease. Am J Physiol Lung Cell Mol Physiol. 2020 Dec 1;319(6):L1021-L1035. doi: 10.1152/ajplung.00207.2020. Epub 2020 Sep 23. PubMed PMID: 32964723; PubMed Central PMCID: PMC7938777.
