Side-by-Side Comparison: COVID-19 Vaccine
This information has been reviewed and approved by Lindsay Sense, RN, BSN, CMSRN (August 2022).
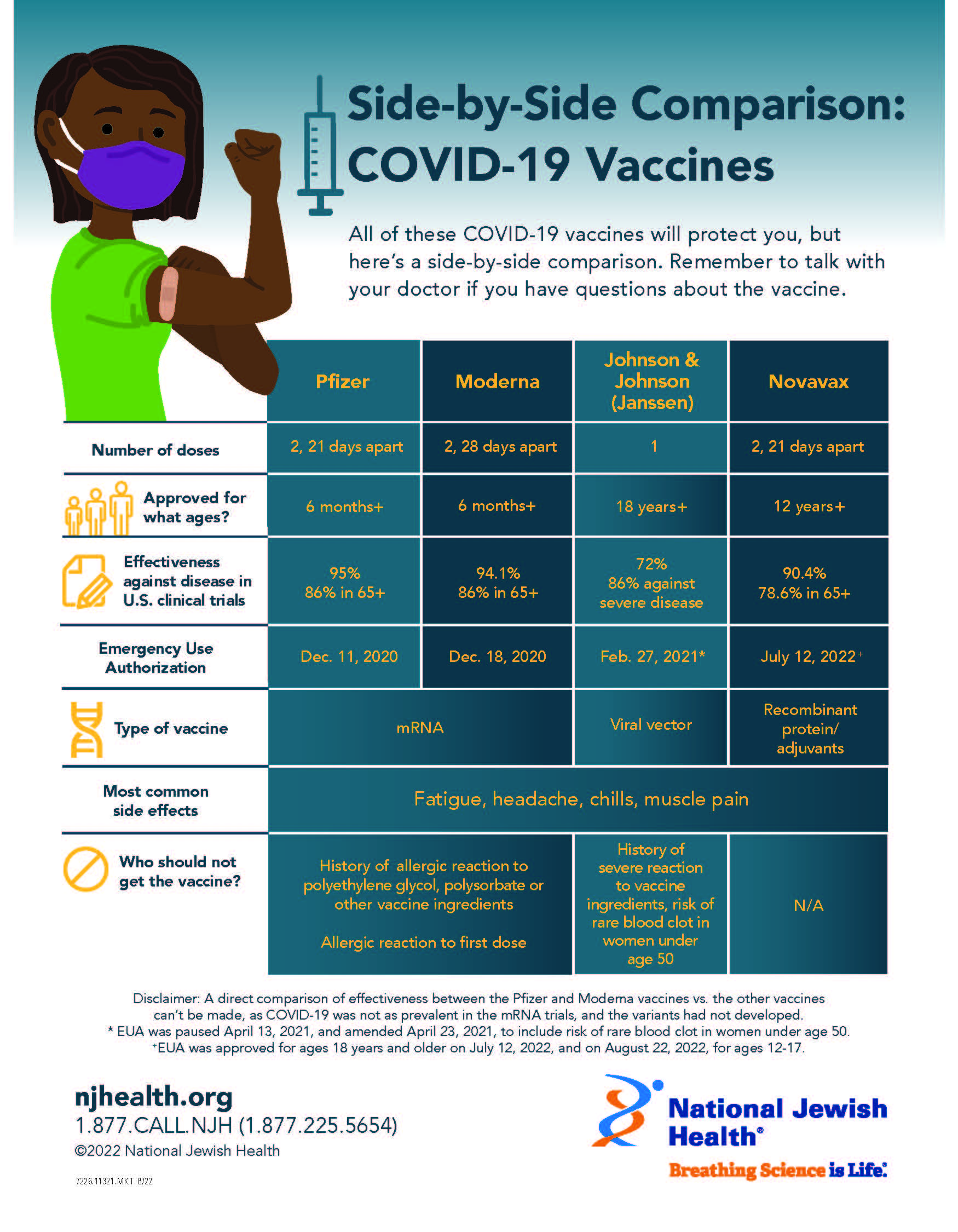
Side-by-Side Comparison: COVID-19 Vaccine
The CDC has advised that, upon receiving the primary doses of one of the approved vaccines, people should also get a booster shot after a specific interval. This is due to the effectiveness of the vaccines waning over time. People who are at-risk for severe infection, such as the immunocompromised, are especially encouraged to get an additional shot. You can read more about booster recommendation details here.
All of these COVID-19 vaccines will protect you, but here’s a side-by-side comparison. Remember to talk with your doctor if you have concerns about getting the vaccine.
| Pfizer | Moderna | Johnson & Johnson (Janssen) | Novavax | |
| Number of doses | 2, 21 days apart | 2, 28 days apart | 1 | 2, 21 days apart |
| Approved for what ages? | 6 months+ | 6 months+ | 18 years+ | 18 years+ |
| Effectiveness against death | Yes | Yes | Yes | Yes |
| Effective against current variants | Yes | Yes | Yes | Yes |
| Effectiveness against disease in U.S. clinical trials | 95% 86% in 65 and older | 94.1% 86% in 65 and older | 72% 86% against severe disease | 90.4% 78.6% in 65+ |
| Emergency Use Authorization | Dec. 11, 2020 | Dec. 18, 2020 | Feb. 27, 2021* | July 12, 2022 |
| Type of Vaccine | mRNA | mRNA | Vector (modified common cold virus) | Recombinant protein/adjuvants |
| Most common side effects | Fatigue, headache, chills, muscle pain | Fatigue, headache, chills, muscle pain | Fatigue, headache, chills, muscle pain | Fatigue, headache, chills, muscle pain |
| Testing for children? | Approved for ages 6 months+ | Approved for ages 6 months+ | TBD | TBD |
| Who should not get the vaccine? | History of allergic reaction to polyethylene glycol, polysorbate or other vaccine ingredients Allergic reaction to first dose | History of allergic reaction to polyethylene glycol, polysorbate or other vaccine ingredients Allergic reaction to first dose | History of severe reaction to vaccine ingredients, risk of rare blood clot in women under age 50 | N/A |
Disclaimer: A direct comparison of effectiveness between the Pfizer and Moderna vaccines vs. the other vaccines
can’t be made, as COVID-19 was not as prevalent in the mRNA trials, and the variants had not developed. * EUA was paused April 13, 2021 and amended April 23, 2021 to include risk of rare blood clot in women under age 50.
+EUA was approved for ages 18 years and older on July 12, 2022, and on August 22, 2022, for ages 12-17.
Additional Resources
Want to use this on your website? Fill out the content usage request form and then copy this code:
| The information on our website is medically reviewed and accurate at the time of publication. Due to the changing nature of the COVID-19 pandemic, information may have since changed. CDC.gov and your state’s health department may offer additional guidance. |


